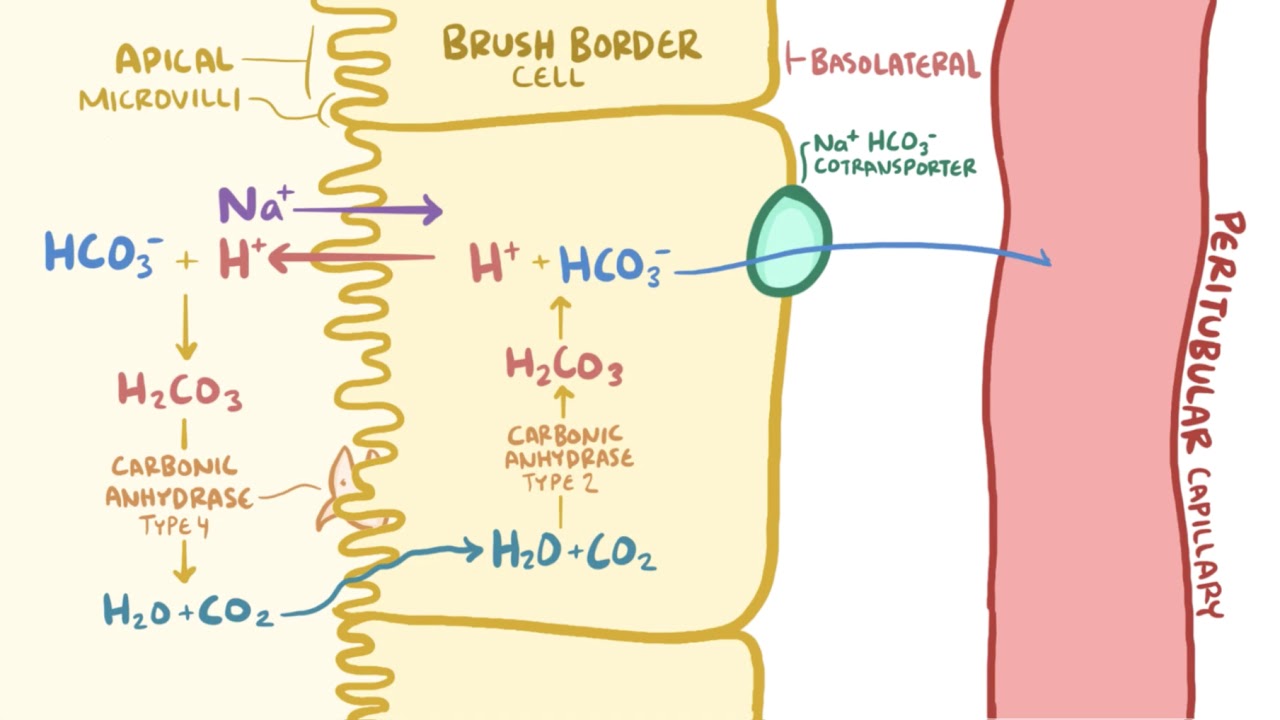
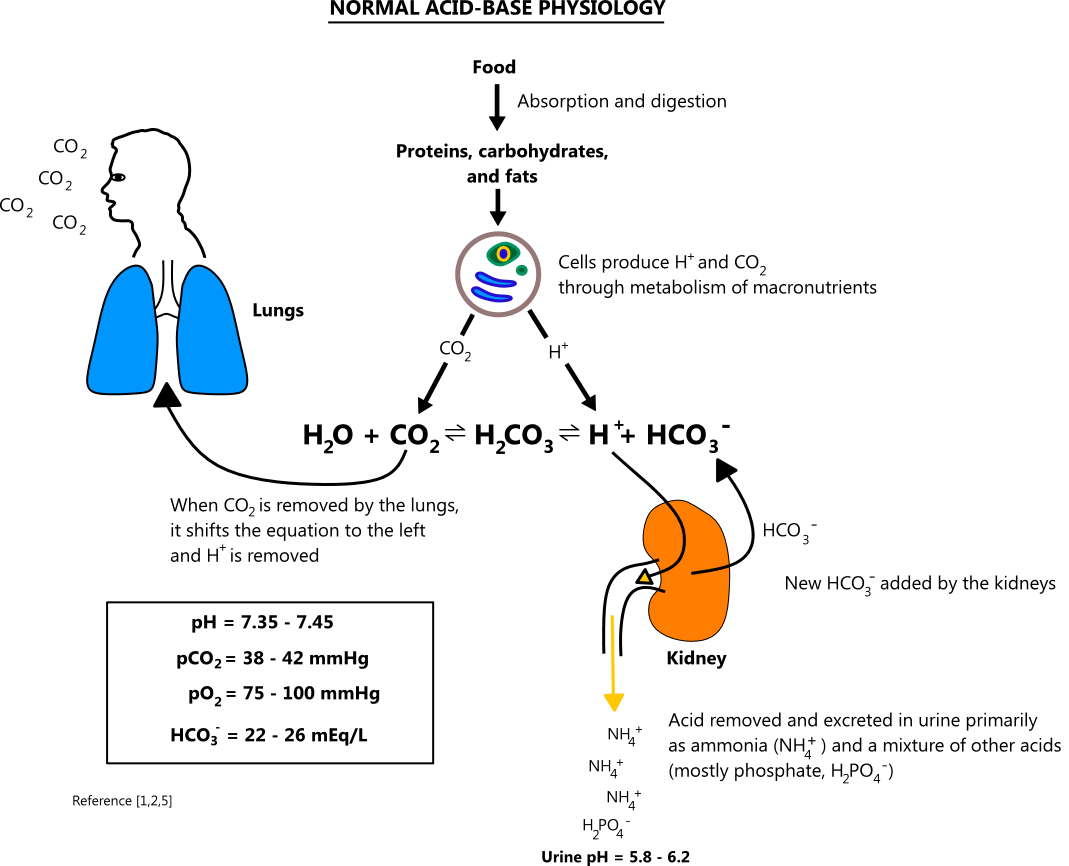
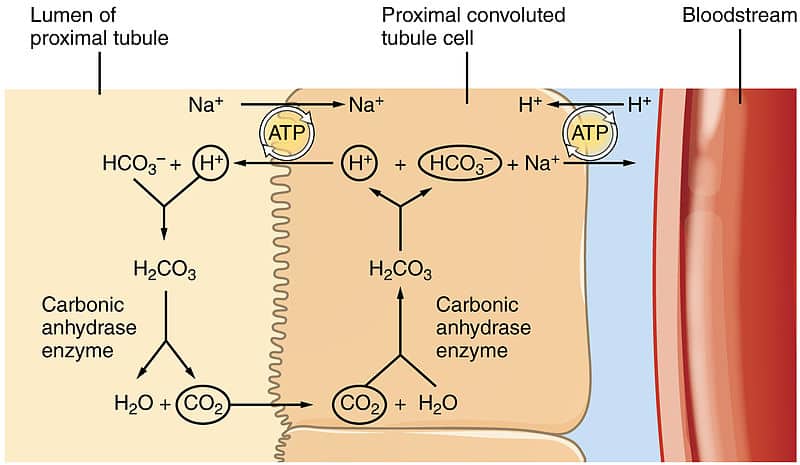
SOLVED: Consider the following reaction. According to Bronsted-Lowry, HCO3- is a(n): HCO3- + H2O —-> H3O+ + CO32- Group of answer choices none of the above base acid conjugate acid conjugate base

Identify the conjugate acid-base pairs in the following reaction. Indicate what each substance is in each pair. H2CO3 + PO43- arrow HCO3- + HPO42- | Homework.Study.com

SOLVED: Konsider the reaction: H2SO3 HCO3" H2CO3 HSO3" a) Identify the acid, base, conjugate acid and conjugate base b. What is the conjugate base of water? C What is the value of [
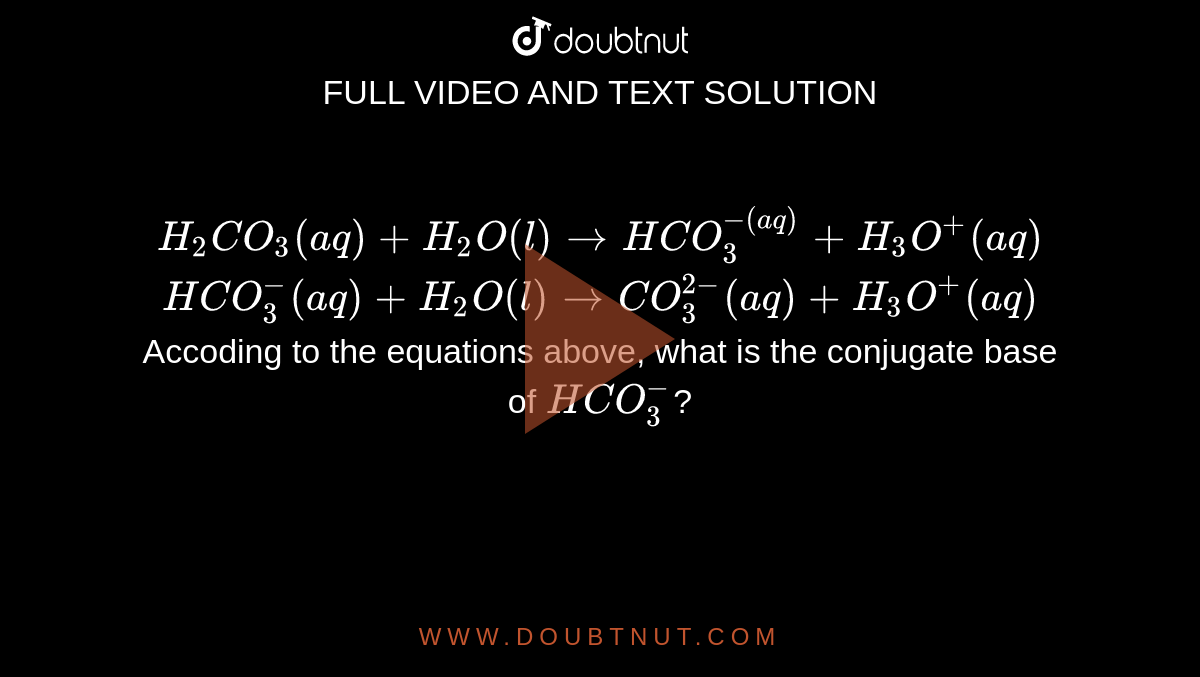
H2CO3(aq)+H2O(l)toHCO3^-(aq) + H3O^+(aq) HCO3^(-)(aq) + H2O(l)to CO3^(2-)(aq)+H3O^(+)(aq) Accoding to the equations above, what is the conjugate base of HCO3^-?
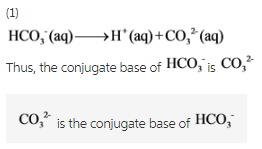
What is the conjugate base of HCO3−? Express your answer as a chemical formula - Home Work Help - Learn CBSE Forum



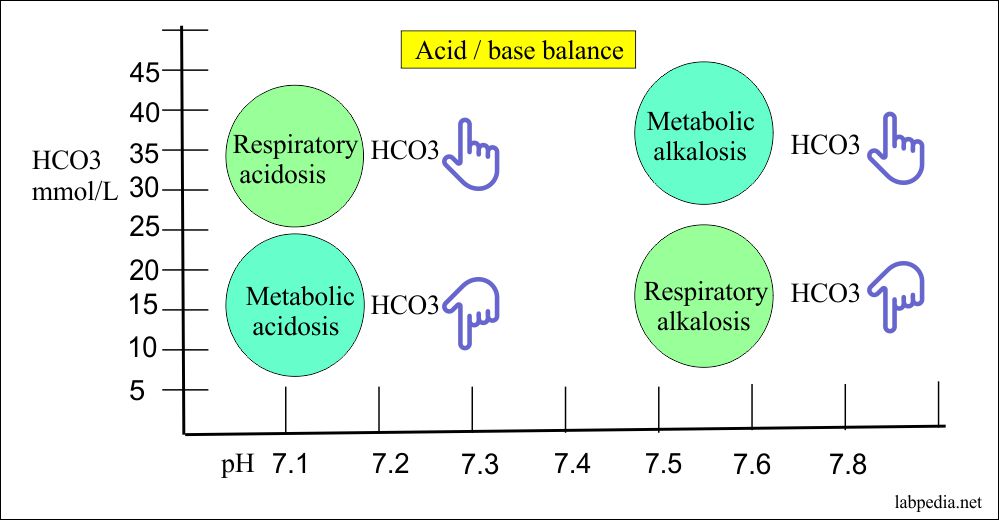

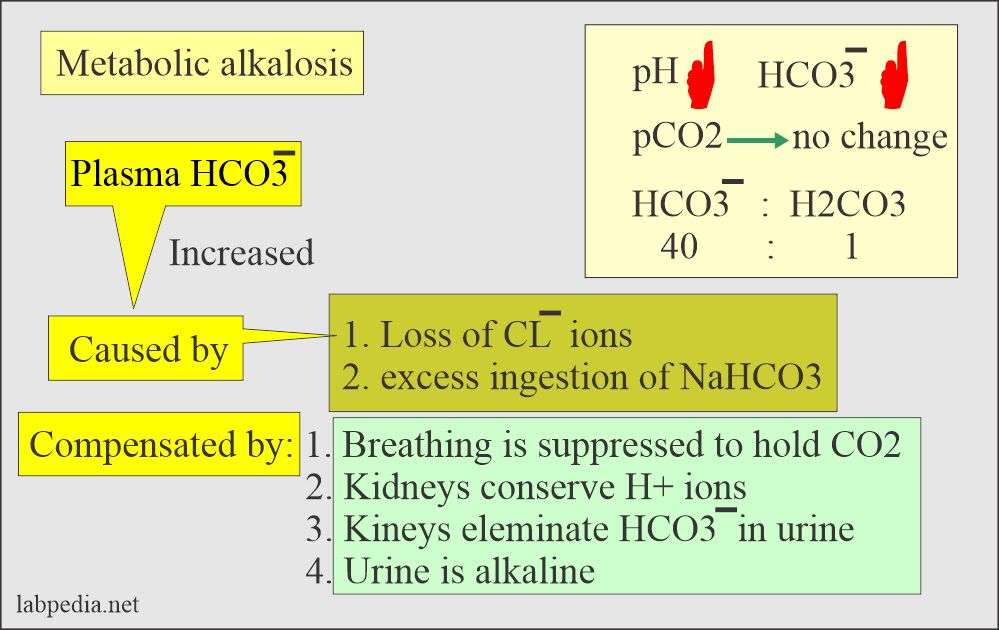
![ANSWERED] In the following reaction: HCO3(aq) + H₂... - Inorganic Chemistry ANSWERED] In the following reaction: HCO3(aq) + H₂... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/53160915-1659276540.7997923.jpeg)